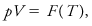Boyle's law
(Also called Boyle–Mariotte law, Mariotte’s law.) The empirical generalization that for many so-called perfect gases, the product of pressure p and volume V is constant in an isothermal process:
where the function F of the temperature T cannot be specified without reference to other laws (e.g., Charles–Gay–Lussac law).