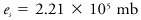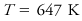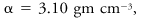critical point
The thermodynamic state in which liquid and gas phases of a substance coexist in equilibrium at the highest possible temperature. At higher temperatures than the critical no liquid phase can exist. For water the critical point is
where es is the saturation vapor pressure of the water vapor, T is the Kelvin temperature, and α the specific volume.