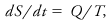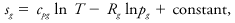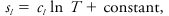entropy
A thermodynamic state variable denoted by S (s denotes specific entropy, entropy per unit mass). The rate of change of entropy of a thermodynamic system is defined as
where Q is the heating rate in a reversible process and T is absolute temperature. Integration of this equation yields the entropy difference between two states. The entropy of an isolated system cannot decrease in any real physical process, which is one statement of the second law of thermodynamics. The specific entropy of an ideal gas, sg, may be expressed as
where cpg is the specific heat at constant pressure of that gas, Rg is its gas constant, and T and pg are its temperature and pressure. The entropy of a liquid sl is
where cl is the specific heat of the liquid.