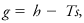Gibbs function
(Also called Gibbs free energy, thermodynamic potential.) A mathematically defined thermodynamic function of state, which is constant during a reversible isobaric–isothermal process. The most important such process in meteorology is the change in phase of water substance. In symbols the Gibbs function g is
where h is specific enthalpy, T is Kelvin temperature, and s is specific entropy.
Compare Helmholtz function.