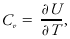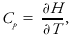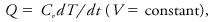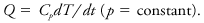heat capacity
(Also called thermal capacity.) The ratio of the energy or enthalpy absorbed (or released) by a system to the corresponding temperature rise (or fall). Heat capacities are defined for particular processes. For a constant volume process,
where U is the internal energy of a system and T is its temperature. For a constant pressure process,
where H is the system enthalpy. The heating rate, Q, for a constant volume process is
whereas in a constant pressure process,